Home » Products » Environment Solutions » Environment Control Systems » Bioquell Biodecontamination Systems
Biodecontamination Systems
Rapid, adaptable and productive
Bioquell Biodecontamination Systems
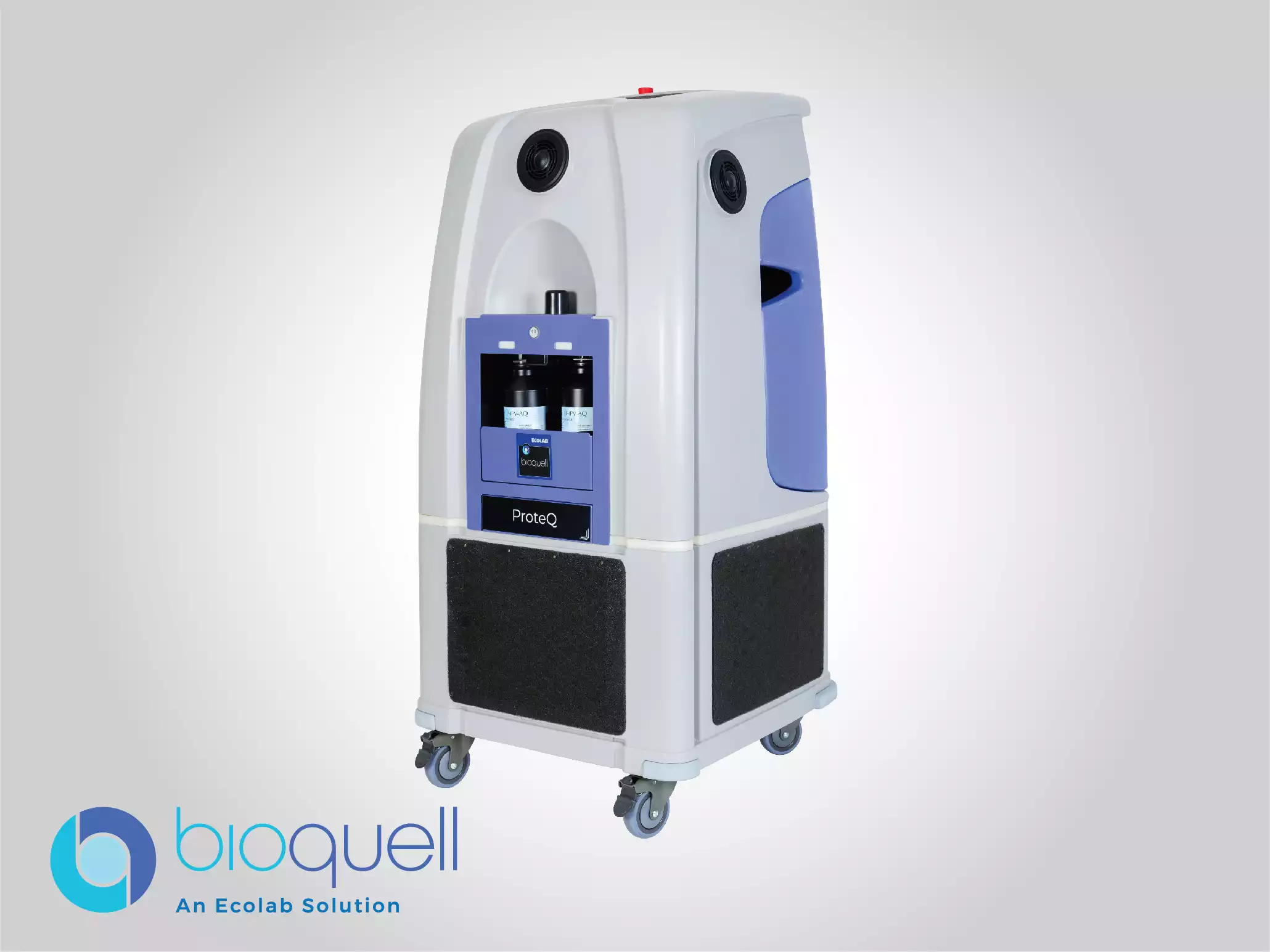
Bioquell ProteQ
Quickly and effectively decontaminate nearly any room or zone with this mobile, scalable and compliant system with wireless communication technology. All components, including built-in aeration and room for additional optional aeration, are hosted within its frame, making setup simple.
Ideal for: Biopharmaceutical Manufacturing Areas, Production Labs, Room and Zone Decontamination, Bio-Safety Labs, Cleanrooms and GMP/GLP Laboratories.
Bioquell Qube
Create an aseptic workspace with Bioquell’s customizable isolator integrated with our Hydrogen Peroxide Vapor technology. Its distinctive design and range of applications far outperform traditional isolators for your decontamination requirements.
Ideal for: Sterility Testing, Gene & Cell Therapy, Small Batch Production, Compounding and Cytotoxic Preparation
Bioquell SeQure
Protect your valuable work from pathogens with Bioquell’s newest decontamination system. Wall-mounted and compact, it eliminates your bioburden with speed and efficiency.
Ideal for: Pass-Throughs & Material Airlocks, Lab & Room Decontamination, Spaces Smaller Than 150 Cubic Meters, Viral & Bacterial Vector Work, Biosecurity and Small Research Areas
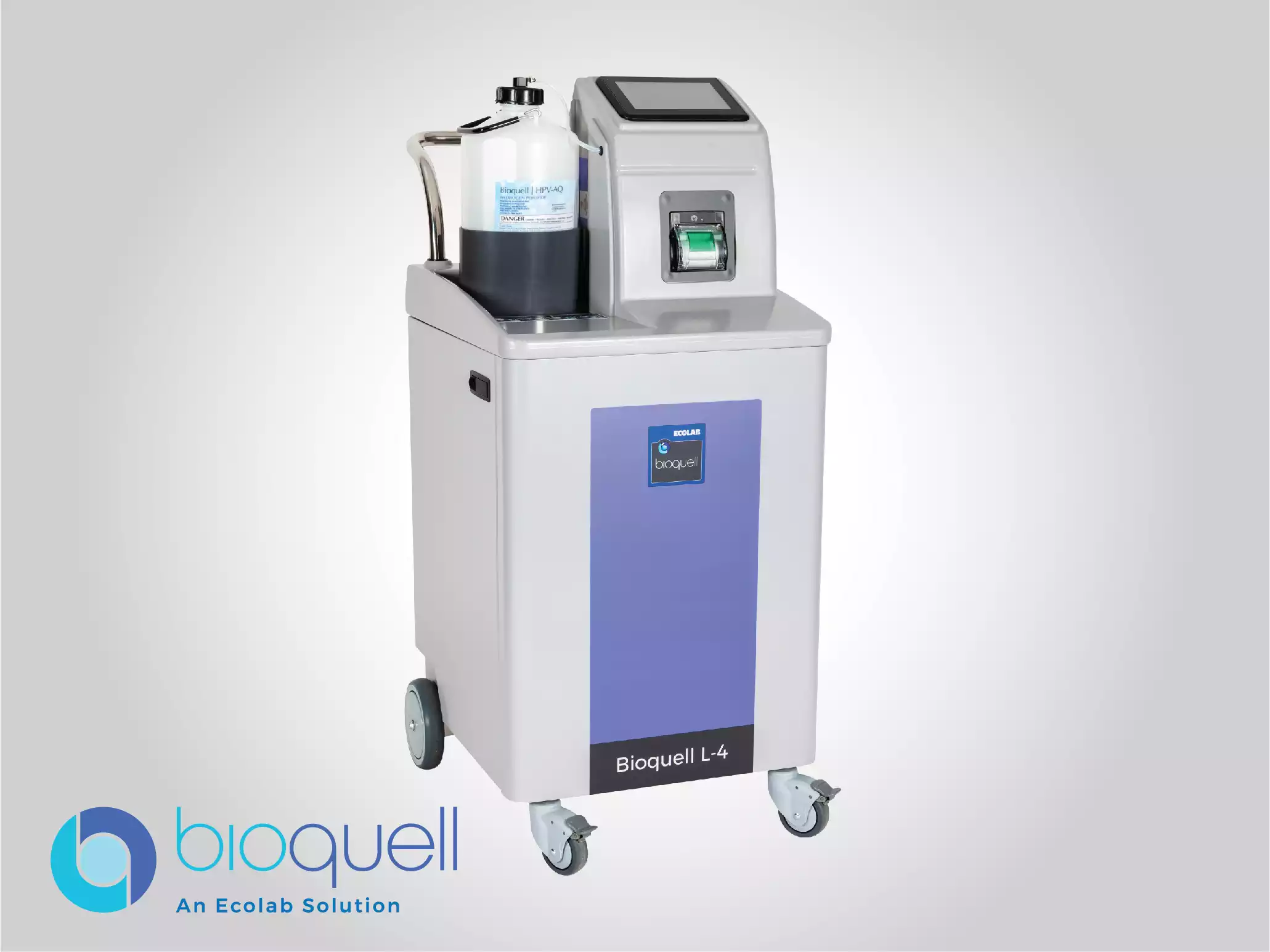
Bioquell L-4
Use Bioquell’s versatile, multipurpose Hydrogen Peroxide Vapor generator to decontaminate surfaces from enclosures and equipment to larger rooms and spaces.
Ideal for: Enclosed Systems & Equipment, Isolators, RABS, Freeze Dryers (Lyophilizers), Bio-Safety Cabinets, Cage & Rack Washers, Incubators, Filter Bank Enclosures and Room Decontamination.
Bioquell IG-2
Make Hydrogen Peroxide Vapor a complete, consistent part of your protocols for total system integration. Ensure the right system set-up with Bioquell’s expert support.
Ideal for: Restricted-Access Barrier Systems (RABS), Filling Lines, Cage and Rack Washers, Isolators, Incubators, Freeze Dryers (Lyophilizers), Robotic Enclosures, Material Airlocks and Small-Volume Rooms.
Bioquell Integrated Building Decontamination System
Create a fully integrated, centralized decontamination solution designed specifically for your facility to reduce manual setup times, increase repeatability, and remove human error.
Ideal for: GMP Biopharmaceutical manufacturing areas, GMP Filling suites, Biomedical facilities and Biosafety/containment laboratories
Read more about Bioquell Biodecontamination Systems